Sexual Behavior Frequency and Menstrual Cycle Length in Mature Premenopausal Women

Winnifred B. Cutler*, Celso R. Garcia +, and Abba Kreiger++
*Department of Biology, University of Pennsylvania, Philadelphia, PA 19104, USA
+ Department of Obstetrics and Gynecology, Hospital of the University of Pennsylvania, Philadelphia, PA 19104 USA
++Department of Statistics, The Wharton School, University of Pennsylvania, Philadelphia, PA 19104 USA. (Received 5 December 1978, in revised form 2. April 1979)
Psychoneuroendocrinology. Vol. 4, pp.297 to 309. c. Pergamon Press Ltd. 1979. Printed in Great Britain
Summary
(1) Women who had regular weekly sexual intercourse with men had menstrual cycles, the average duration of which was 29 days (range 26-33), while women with less frequent behavioral interactions tended to have more extreme cycle lengths, as shown in 3 different studies in which prospective double blind measurements of data were used.
(2) The effect was noted consistently in college women but also occurred in infertile patients. Sexual behavior and consistent menstrual cycle length were associated in all women studied.
(3) The mechanism appeared to be related to the regularity with which sexual activity occurred during all the non-menstruating days of the cycles.
Introduction
The demonstration that changes in mammalian sexual behavior frequently parallel changes in female endocrine status has led to a variety of experiments designed to investigate cause and effect. The relations appear to be circular as shown by experiments that manipulate either variable. In non-human experiments the findings are clear. Whether naturally occurring or experimentally induced, changes in endocrine state effect subsequent sexual behavior in rat (Weizenbaum, 1976; Zemlan & Adler, 1977), dog (Hart, 1970) and rhesus monkey (Michael & Keverne, 1970; Michael, Keverne & Bonsall, 1971; Anderson & Mason, 1977). Likewise, the influence of sexual behavior on endocrine state has been established for the rodent (Adler, 1969; Moss & Cooper, 1973). The case is less clear for humans where experimental results have been mixed. A delayed age at first coitus was noted among infertility patients where those who had been unable to conceive showed the latest first coital ages (20.47 yr); habitual aborters, somewhat late first coital ages (19.11 yr); and routine gynecology patients, earlier first coital ages (18.33 yr) which were comparable to population norms (Cutler, Garcia, Krieger, 1979). Endocrinologists have reported changing levels of coital frequency which parallel, and were presumed to follow, hormonal variations (Udry & Morris, 1968), but subsequent studies failed to confirm this (Morris & Udry, 1977). The seeming ambiguity may have been resolved with the recent demonstration of increases in female initiated sexual activity in contrast to total sexual activity at the presumed ovulatory time of the cycle (Adams, Gold & Burt, 1978). Thus, the failure to demonstrate a mid-cycle behavioral change appears to have resulted from inadequate resolution of the relevant components of sexual behavior. Studies of sexual behavior in the human have failed to provide any post-coital LH response in men or women (Stearns, Winter & Faiman, 1973; Davidson & Trupin, 1975): although an LH surge induced by an erotic film has been reported for men (LaFerla, Anderson & Schalch, 1976). The possibility that humans might, under some circumstances, show a low incidence of reflex ovulation during the follicular phase of the cycle was suggested by Jochle (1975) using the older German anecdotal literature. Since positive confirmation has not been reported in the last 20 yr, it seems more prudent to consider the ideas as intriguing but unsubstantiated.
In summary, human studies to date have failed to show the clear reproductive system effect resulting from sexual behavior that has been shown for other mammals. The possibility that sexual behavior patterns themselves could be related to menstrual cycle pattern prompted this series of investigations. The questions being posed are: (1) is menstrual cycle length related to frequency of sexual behavior? And: (2) if so, what types of sexual behavior are associated with different cycle lengths?
Methods
In the present study sexual behavior frequency was measured in terms of its occurrence within the time span of a week rather than simply total number of events per cycle. Subjects who showed a minimum weekly incidence of coitus ('weekly') are compared with those reporting a frequency of less than once a week ('sporadic' or 'never'). This report consists of 3 substudies investigating the possibility of an association between coital frequency and menstrual cycle length. The 1st substudy, which was conducted in the Fall of 1976, recruited university co-eds and served as a preliminary report. It relied on retrospective data of coital frequency (prospective menstrual onset data). The 2nd substudy was a more structured replication of the first. It employed a similar, but 4 times larger sample and used prospective collection of all relevant data. Substudy 3 retrospectively collected prospectively recorded menstrual and coital information from gynecological records which had been maintained by the infertility patients of one author (CRG). Informed consent by the students and university approval of the details for the research design were obtained before data collection began. Confidentiality was assured in all studies by coding all data to provide anonymity.
Substudy 1. Prospective menstrual data retrospective sexual data (double blind)
In a preliminary study, undertaken in the Fall of 1976, 60 female students (aged 19-22 yr) attending the University of Pennsylvania kept a prospective record of their menstrual cycles on specially provided calendars. Three female, undergraduate contemporaries of the subjects served as data collectors and also recruited participants by knocking on dorm doors and presenting the aims of the study. * Criteria for participation required that subjects be gynecologically mature (Treloar, Boynton, Behn & Brown, 1967) (minimum 7 yr since menarche), non-users of any hormone drug (such as birth control pill) and non-users of the intrauterine device (IUD) which might have affected the pattern of uterine bleedings, as suggested by Jones, Sabry & Hodgson (1974) and Mayes (1977). The study began in early September and ended with the collection of data cards in mid-December. In a personal interview with the data collector in November 1976, blood pressure was monitored and information on a variety of behavioral assessments, including self-assessment of autumn sexual intercourse frequency rated as weekly, sporadic or never, was obtained.
*A study designed to investigate menstrual cycle patterns and other behavioral and physiological measures.
Substudy 2. Prospective menstrual and sexual data (double blind)
In the Fall of 1977, 312 University of Pennsylvania students ranging in age from 19 to 22 yr. were recruited by 13 female undergraduate contemporaries who knocked on randomly pre-assigned dorm doors to solicit participation. Participation in the study required that subjects meet all the criteria of the preliminary study with respect to age, gynecological maturity and contraceptive method. Subjects were additionally required to be unmarried, nulliparous, white and to have completed a minimum of one full academic year. The data collectors were not permitted to recruit more than one subject per dormitory room. Four experimental groups were formed (1A, 2, 3, and 1B). In each group the data collectors were trained to solicit subjects, conduct interviews and administer blood pressure and prepackaged psychological tests. The data pool was flooded with more bits of information than were deemed relevant in order to assure a double blind situation in which the research objective was masked. Dilution was used to decrease the risk that the participants would supply information in conformance with their expectation of what were appropriate answers (Schacter & Singer, 1962). A specially prepared printed menses calendar was distributed to all students for prospective recording of their individual days of menstrual flow. The sexual data were surveyed in three ways: (1) Group 1A: 4 data collectors monitored 101 students. Weekly phone calls were made to each student to determine and record coital days of the previous week on a calendar maintained by t he data collector. (2) Group 2: 3 data collectors monitored 68 students. Group 3: 3 data collectors monitored 79 students. These 147 students all recorded days of coitus © defined as penile insertion into the vagina; as well as days in which they experienced genital stimulation (GS) by a male partner, short of penile insertion. Group 3 students in addition recorded days of masturbation (M). Appropriate behavior calendars were provided with the menses calendars. (3) Group 1B: 3 data collectors 64 students. This was a replication of Substudy 1., Fall 1976, in which prospective menses data were combined with retrospective estimate of coital frequency.
In mid-October, a 15 min interview was held with each student, ostensibly to survey a range of behavioral and physiological measures including blood pressure, psychosomatic and generalized personality inventories. For groups 1B, 2, and 3 follow-up reminders to continue recording were transmitted every 3 weeks to each subject by her data collector. Group 1A subjects were contacted weekly by phone. ,A final interview at the end of the study (following the last menstrual onset after December 1, 1977, or by December 15 if no onset appeared) consisted of a written document of ten additional behavioral survey questions for groups 1A, 2, and 3. All documents were sealed in the presence of the experimenter and the subject and mailed to one of the authors' (WBC) laboratories. For group 1A, the name and phone number heading on the coital calendar was removed before sealing and mailing the now anonymous document. Each data collector began the study with 30 students. On average 5 subjects were lost from the study; this tended to be due to subjects starting oral contraceptives.
Substudy 3. Prospective menstrual and coital data: gynecological records
A review of the active files (patients seen within the last 2yr) of the infertility-gynecological practice of one author (DRG) produced 192 patient files which contained basal body temperature charts. Careful scrutiny of these yielded 60 usable files which met the following minimum criteria: legible data for at least one menstrual cycle defined by the recording of its 2 catamenial limits; coital data legibly recorded on chart; attributable date of the recordings which then permitted confirmation that no drugs were ingested for the period of tabulation as well as during the preceeding 3 months and absence of endometrial biopsy during the time of recording tabulations. These 60 patients presented a variety of infertility diagnoses which included occlusion of the fallopian tubes, polycicstic ovaries, pelvic adhesions, habitual abortions, male factor (spermatic pathology), cervicitis and primary infertility with no detectable pathology. They arranged in age from 20 to 41 yr as shown in Fig. 7. All were attempting conception and most were nulliparous.
Method of data analysis (all studies)
The selection of the Segal-Tukey test provided a highly appropriate method of analysis for this type of research (Lehman, 1975). Weekly sexual behavior defined the experimental condition, less than weekly (sporadic and never) defined the control condition and cycle length was the variable being tested ('score'). A rank ordering of scores permitted one to test whether different conditions showed a different range of scores. One was therefore measuring relative variance of scores from the two conditions. Selection of one cycle per subject was deemed necessary because: (1) there was no adequate way of dealing with different numbers of cycles per person since subjects provided 1, 2, 3 or 4 menses onsets during the fall semester and (2) observations from any one individual had a degree of dependency. The first cycle was selected for all figures but statistical analyses showed that regardless of whether one tested first, last or mean cycle lengths, the results were very similar. Unfortunately only mean data were available for substudy 1. Determination of frequency of behavior for university subjects involved inspection of their behavior data charts covering the span of the semester. Weekly categorization required a report of sexual behavior every week of the term and thus reflected an ongoing pattern of weekly behavior. For Substudy 3, the infertile sample, in which different time spans of record were available for different patients, consistency required behavior categorization to span only the one cycle under consideration, ie. the first one.
Results
Substudy 1. Prospective menstrual data and retrospective coital data
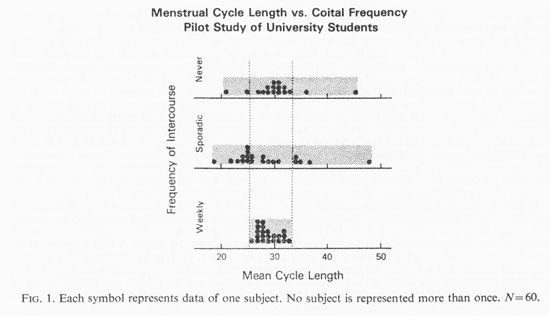
Each student's mean cycle length during the fall semester is shown in Fig. 1 grouped by her retrospective estimate of coital frequency category. The weekly subjects displayed a cluster of menstrual events within the 26-33 day range (mean 29.5). Less frequent sexual activity was associated with short, medium, and long cycles which were significantly more variable in length than those of the former group (z = 3.21, p <0.001). A variety of personality factors showed no association with either cycle length or sexual behavior. The following were investigated but the associations did not appear: incidence of depression or monthly cramps; variability of the cycle; systolic and diastolic blood pressure levels; liability of blood pressure and measures of neuroticism, introversion and extraversion (Eysenck, 1975).
Substudy 2. Prospective menstrual and sexual data
Figure 2 shows the first cycle of all 248 students grouped by coital frequency. Again the weekly subjects displayed a relative centering of cycle lengths about 29.5 days (z= 1.88, p<0.03) while less than weekly behavior was associated with more extreme cycle lengths. To evaluate whether this effect was associated with short and/or long cycles, two individual Wilcoxon Rank tests were applied removing the data occurring at the median from the analysis, at the suggestion of Dr. John Tukey (personal communication). This showed that the association between minimum weekly coital behavior and fewer extreme cycle lengths existed for both short and long cycles with a greater reduction of the short cycles (short z = 1.60, p <0.05; long z = 1.34, p <0.09). Included in the weekly category (Fig. 2) were those women whose coital frequency was at least twice a week. The median of these 248 cycle lengths was 29 days with 22 people having this length. Figure 3 shows the first cycle of the 147 students for whom genital stimulation (GS) data as well as coital information was available. This includes experimental groups 2 and 3. The 101 subjects of group 1A were eliminated from this analysis as only weekly coital information had been obtained by telephone. Again, weekly subjects tended to cycle at lengths approximating 29.5 days. Less frequent behavior was associated with a wider range of short, medium and long cycles (z = 2.29, p 0.01). A stronger effect toward fewer of the short cycles was again noted (z=1.73, p <0.04); while a possible trend existed for the reduction in numbers of long cycles in association with weekly sexual activity (z=1.07, p <0.14).
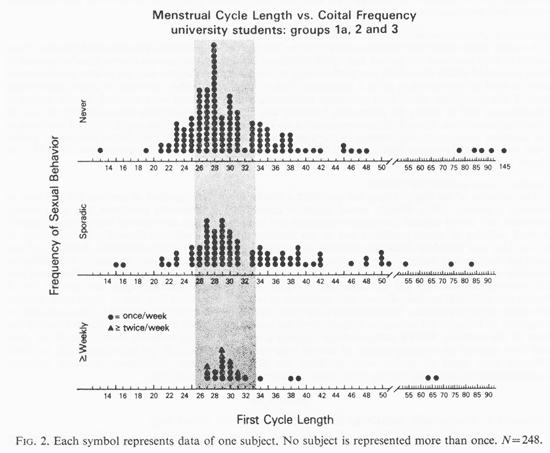
The symbols in Fig. 3 show which of the less than weekly subjects (box) were newly added to the weekly groupings by the inclusion of GS data. Inspection supports the statistical impression of a stronger effect for diminished number of short cycles in association with weekly behavior.
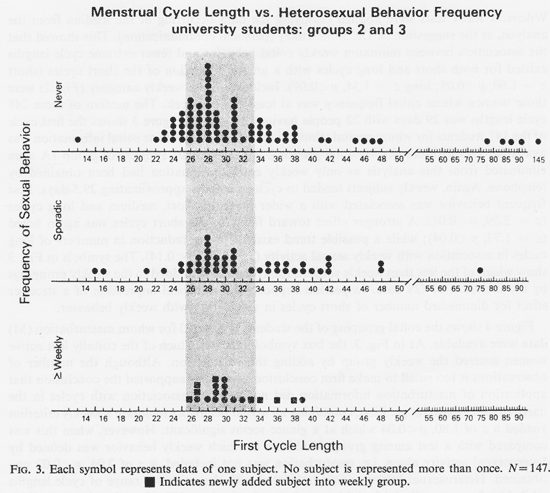
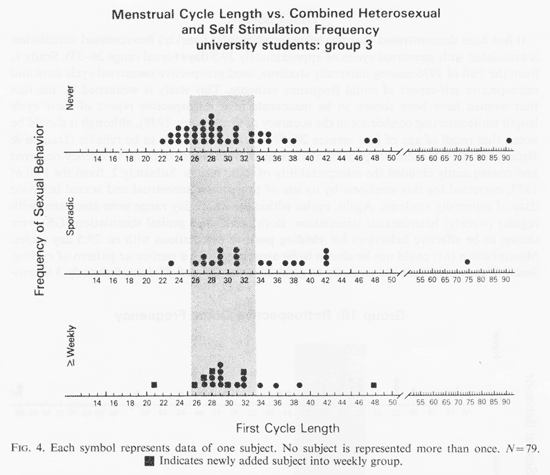
Figure 4 shows the coital grouping of the students in group 3 for whom masturbation (M) data were available. As in Fig. 3, the box symbol indicates which of the coitally less active women entered the weekly group by adding the M criterion. Although the number of observations is too small to make firm conclusions, two testes supported the conclusion that application of masturbation information did not yield an association with cycles in the narrow (ca. 29 days) range. First, combining all sexual behaviors to obtain a weekly criterion yielded a z of 1.80, p<0.04 which at a glance seems significant. However, when this was compared with a test among group 3 subjects in which weekly behavior was defined by heterosexual activity alone, i.e masturbation was not included, a z of 2.04, p<0.02 was obtained. Heterosexual behavior was associated with the narrower range of cycle lengths while M information diminished this association. Inspection of Fig. 4, noting the symbolically indicated weekly subjects employing the M criterion, shows that the few masturbation additions to the weekly group were heavily weighted by the larger proportion of heterosexual weekly activity points. This, both by data inspection and by statistical evaluation the M criterion seemed inadequate as an associate of particular cycle lengths. A similar analysis applied to Fig. 3 shows that the addition of GS data did not distort the weekly coital picture. The z score without GS data for those 147 students was less without the GS weekly points (z= 1.92, p <0.03). If anything could be reasonably inferred about these small N's, it would be that GS data improved the result.
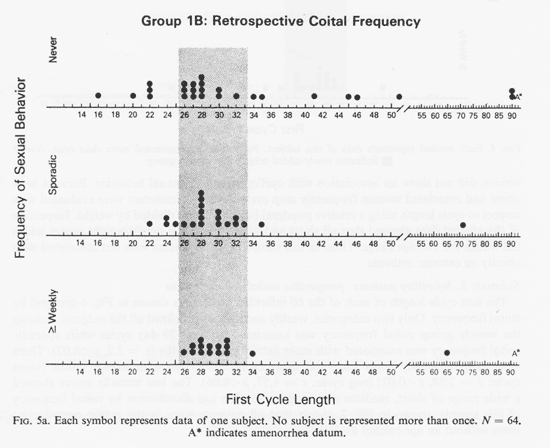
Two replications of substudy 1 are now presented: (1) Group 1B was employed as a control to replicate the 1976 work. Figure 5a shows the first cycle of these prospective cycling reports grouped by each subjects' retrospective estimate of her coital frequency. Minimum weekly coital behavior was associated with cycles in the ca. 29 day range (z= 2.11, p<0.02). (2) Mean cycle lengths of the groups 1A, 2 and 3 subjects are presented in Fig. 5b.
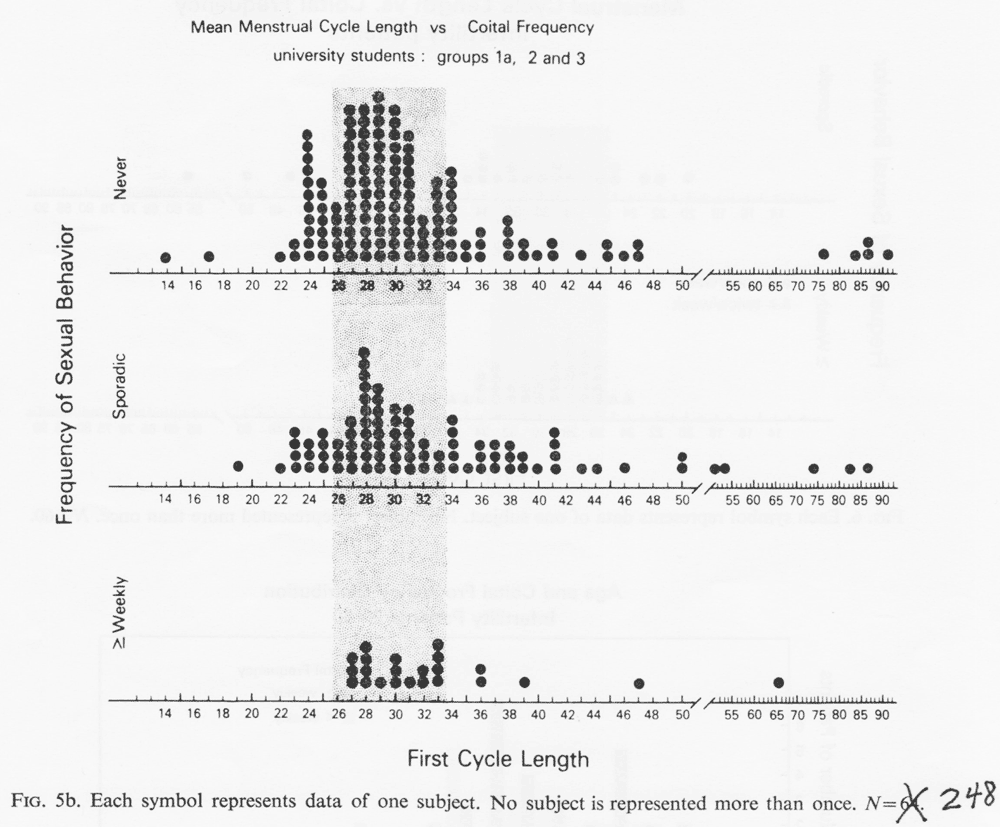
They also show the same pattern of fewer extreme cycle lengths among the weekly subjects. As in substudy 1, cycle variance, neuroticism, extraversion and intraversion did not show an association with cycling length or sexual behavior. Because both obese and emaciated women frequently stop cycling, these parameters were evaluated with respect to cycle length using a relative ponderal index of height divided by weight. Inspection of the plotted data showed that all short and long cyclers had height/weight scores which fell within the average range. Thus, in this sample, extreme cycles were not associated with obesity or extreme esthenia.
Substudy 3. Infertility patients: prospective coital and cycle data
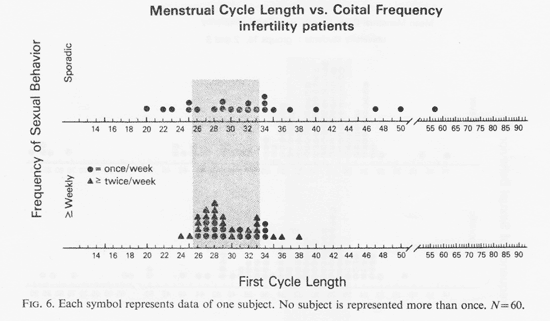
The first cycle length of each of the 60 infertility patients is shown in Fig. 6 grouped by coital frequency. Only two categories, weekly and sporadic, defined all the subjects. Among the weekly group coital frequency was associated with more extreme cycle lengths (z = 2.2, p <0.02). There were proportionately fewer long and short cycles associated with weekly coitus (short cycle: z = 2.84, p <0.02; long cycle: z = 1.57, p<0.06). The less sexually active showed a wide range of short, medium and long cycles. The age distribution by coital frequency of this sample, shown in Fig. 7, shows that all patients were in the active reproductive years without an age-related association.
Discussion
It has been demonstrated that the behavior of regular (weekly) heterosexual stimulation is associated with menstrual cycles of approximately 29.5 days (usual range 26-33). Study 1, from the Fall of 1976 among university students, used prospective menstrual cycle data and retrospective self-report of coital frequency estimate. This study is weakened by the fact that women have been shown to be inaccurate in a retrospective report of their cycle length while asserting confidence in the accuracy of same (Arey, 1939), although it should be noted that recall of age of first menses 20-40 yr later was shown to be reliable (Damon & Bajima, 1974). It is therefore possible that an inaccurate report of coital frequency occurred and consequently clouded the interpretability of these results. Substudy 2, from the Fall of 1977, corrected this weakness by its use of prospective menstrual and sexual behavior data of university students. Again, cycles within the ca. 29 day range were associated with regular (weekly) heterosexual stimulation. Both coitus and genital stimulation (GS) were shown to be effective behaviors for yielding positive associations with ca. 29.5 day cycles. Masturbation (M) could not be shown to be associated with a particular pattern of cycling. Small N's severely limit the capacity to generalize the GS and M data. Substudy 3 investigated a sample of infertile women who prospectively had maintained coital and menstrual data incidental to keeping body temperature charts. Again, cycles within the approximate 29 day range showed a definite association with regular (weekly) sexual behavior. Regular coital stimulation was also investigated to see if twice weekly behavior was associated with a different pattern of cyclicity than weekly behavior. The results were mixed and since the N's were small no definite conclusion is suggested. The possibility that sporadic sexual activity might be due to the practice of the rhythm method of birth control was considered. A careful review of each behavior chart of each sporadically active woman showed only two people who avoided intercourse in the presumed pre-ovulatory range (14-21 days premenstrually). As these two patients had ca. 29 day cycles, t heir absence from statistical evaluation would not distort the results. Lest the reader, at first glance, consider the sexual activity of the university women unusually low it should be remembered that the conditions of this study prevented many sexually active women from participation. The use of either an IUD or a contraceptive hormone drug was sufficient condition to bar participation. Because of the need for privacy, we could not obtain data on the reason why about half of the women approached expressed reluctance at having to disqualify themselves once the list of criteria for participation was read.
Since others have suggested that copulation the week before ovulation facilitates ovulation, as described in the introduction, it is interesting to note that our data do not support this hypothesis. In fact, inspection of these data showed that the infertile women tended to clump coital events at presumed preovulatory days in those instances where sporadic coital activity occurred. The sporadic college women showed activity at the presumed ovulatory time of the cycle. This could explain why investigators have repeatedly been unable to demonstrate any of the components of reflex ovulation. The extremes of cycle lengths are an expression of a reduced fertile capacity would seem to be quite clear. Two types of data lead to this conclusion: (1) cycle length evaluations, especially with respect to basal body temperature analysis and (2) physiological studies in humans and rhesus monkeys which show various endocrine states being reflected in length of cycle or length of one of the phases of the cycle. It has long been known that some human 'menstrual' bleedings are independent of ovulation; although in many instances the bleeding is superficially indistinguishable from the menstrual bleeding of a regular ovulatory cycle (Hartman, 1931; Corner, 1933; Garcia & Rock, 1958; Garcia & Rosenfeld, 1977).
Recently, Vollman (1977) demonstrated that the incidence of anovulatory cycles increases as a function of increasing extremes in cycle lengths. Earlier work showed similar data (Collet, Wertenberger & Fiske 1954). Vollman's' prospective measurements showed that the incidence of monophasic temperature charts increased with increasing extremes of cycle lengths; and that monophasic temperature charts are characteristic of men, pre-pubertal girls and postmenopausal women. Biphasic temperature charts, in contrast, are known to be characteristic of ovulatory cycles (Hartman, 1972; Vollman, 1977; Garcia & Rosenfeld, 1977). Studies of infertile populations have shown that infertile women tend to have extreme menstrual cycles, either short or long (Saxena, Poshyachinda & Dusitin, 1976; Gautray, Jolivet, Goldenberg, Tajchner & Eberhard, 1978). From Vollmans' very large sample studies representative figures indicate the degree of monophasic incidence with cycle length variation: in short cycles of 7-17 days, over 57% of the charts are monophasic (anovulatory); in cycles greater than 51 days, 40% are monopahsic. In contrast, fewer than 2% of the temperature charts are monopahsic in cycles with lengths between 26 and 33 days. One may conclude that cycles of extreme lengths tend to be anovulatory and are infertile while cycles of 26-33 days tend to be ovulatory and potentially fertile.
In the college women there were not short cycles among the coitally active (weekly) women; and there were fewer long cycles among these than in the less active women. The infertile population showed this trend also. There were fewer short and long cycles among the sexually active although one 24 day and one 25 day cycle within this group were noted. A review of the physiology-endocrine studies may clarify these results. Current evidence suggests that very short cycles reflect a greater potential for infertility in that cycle while long cycles represent only relatively reduced fertility: (1) Cycles shorter than 26 days may represent abortive follicular or luteal functions since the proliferative phase appears to require 12.5 days for follicular development and the secretory phase is established to be relatively fixed at 16 + 2 days in humans (Tsai & Yen, 1971; Mishell, Nakamura, Grosignani, Stone, Kharma, Nagata & Thorneycroft, 1971; Cargille, Vaitukaitis, Bermudez & Ross, 1973; Korneman & Sherman, 1973) and monkeys (Niswender & Spies, 1973; Baird, Baker, McNatty & Neal, 1975; Goodman, Nixon, Johnson & Hodgen, 1977). Thus 26 days seems to be the usual minimum ovulatory cycle length. (2) Long cycles may reflect subnormal function and, therefore, sterility as suggested by data from humans (Tsai & Yen, 1971) and monkeys (Baird et al., 1975). Alternatively the long cycler may be fertile each cycle but simply have fewer fertile days per year. It is therefore reasonable to conclude that, with rare exceptions, all the subjects showing short cycles and some of the subjects with long cycles are infertile during that cycle.
Since infrequent sexual activity is associated with an increased likelihood of extreme cycle length and extreme cycle lengths tend to be anovulatory, we take the view that infrequent sexual activity and infertility may be associated. The direction of causality cannot be deducted from these data, and this would be an interesting subject for future investigations.
References
Adams, D.B., Gold, A.R. & Burt, A.D. (1978) Rise in female-initiatined sexual activity at ovulation and its suppression by oral contraceptives. New Engl. J. Med. 299, 1145-11150.
Adler, N.T. (1969) Effects of the male copulatory behavior on successful pregnancy of the female rat. J. comp. physiol. Psychol. 69, 613-622
Anderson, C.O. & Mason, W.A. (1977) Hormones and social behavior of squirrel monkeys. Horm. Behav. 8,100-106
Arey, L. B., (1939) The degree of normal menstrual irregularity. Am. J. Obstet. Gynec. 37, 12-29
Baird, D.T., Baker, T. McNatty, K.P., & Neal, P. (1975) Relationship between the secretion of the corpus luteum and the length of the follicular phase of the ovarian cycle. J. Reprod. fert. 45, 611-619
Cargille, C.M. Vaiatukatitis, J.L. Bermudez, J. A. & Ross, G. T. (1973) A differential effect of ethineyl estradiol (EE) upon plasma FSH and LH relating to time of administration in the menstrual cycle. J. clin. Endocr. Metab. 36, 87-94.
Collet, M. E., Wertenberger, Q.E. & Fiske, V.M. (1954) The effect of age upon the pattern of the menstrual cycle. Fert. Steril 5, 437-448
Cutler, W.B., Garcia,
C.R. & Krieger, A.M. (1979) Infertility and age at first coitus. J. Biosoc. Sci. (in press)
Damon A. & Bajima, C.J (1974) Age at first menarche: accuracy of recall after thirty-nine years. Hum Biol. 46, 381-384
Davison, J.M. & Trupin, S. (1975) Neural mediation of steroid-induced sexual behavior in rats. In Sexual Behavior Pharmacology and Biochemistry, M Sanadler and G.L Gessa (Eds.), pp 13-20. Raven, New York
Eysenck, H.J. (1975) The measurement of emotion. In 'Emotions' Their Parameters and Measurement. L. Levi (Ed.), 437-467. Raven, New York.
Garcia, C.R. & Rock, J. (1958) Ovulation. In Essentials of Human Reproduction. J. Valardo (Ed.) pp. 22-54. Oxford University Press, New York.
Garcia, C.R. & Rosenfeld, D. (1977), Human Fertility, The Regulation of Reproduction. F.A. Davis, 69.
Gautray, J. P., Jolivet, A. Goldenberg, F., Tajchenr, G. & Eberhard, A. (1978). Clinical investigation of the menstrual cycle II. neuroendocrine investigation and therapy of the inadequate luteal phase. Fert. Steril. 29, 275-281
Goodman, A.L. Nixon, W.E., Johnson D.K. & Hodgen, G.D., (1977) Regulation of folliculogenesis in the cycling rhesus monkey: selection of the dominant follicle. Endocrinology 100, 155-161
Hart, B.L. (1970) Mating behavior in the female dog and the effects of estrogen on sexual reflexes. Horm Behav. 1, 93-104
Hartman, C.G. (1931) On the relative sterility of the adolescent organism. Science 74, 226-227
Hartman, C.G. (1972) Science and the Safe Period. Kreiger Publishing Company, New York.
Jochle, W. (1975) Current research in coitus-induced ovulation; a review. J. Reprod. Fert. Suppl 22, 165-207
Jones, D.J. Sabry, A. & Hodgson, B.J> (1974) Effect of IUDs on uterine cyclic AMP in rabbits. Live Sci. 14, 1577.
Korenman, S. & Sherman, B. (1973) Measurement of serum LH, FSH, estradiol, and progesterone in disorders of the human menstrual cycle: the inadequate luteal phase., J. clin Endocr. Metab. 36, 1204-1209.
LaFerla, J.J., Anderson, D. L., Schalch, D.S., (1976) Psychoneuroendocrine response to visual erotic stimulation in human males. Psychosom. Med. 38, (1) 62.
Lehman, E.L. (1975) Nonparametric Staistical Methods Based on Ranks. Holden Day, San Francisco.
Mayes, D. 1977) A comparison of menstrual bleeding patterns associated with three IUD models: an example of reference period analysis. J. Biosoc. Sci. 9, 121-135
Michael, R.P> & Keverne, E.B. (1970) Primate sex pheromones of vaginal origin. Nature 225, 84.
Michael, R. P. & Keverne, E.B. & Bonsall, R.W. (1971) Pheromones: Isolation of male sex attractants from a female primate. Science 172, 964.
Mishell, D.R., Nakamura, R. M., Grosiganin, P.G., Stone, S., Kharma, K., Nagata, Y. & Thorneycroft, I. (1971) Serum gonadotropin and steroid patterns during the normal menstrual cycle. Am. J. Obstet., Gynec. 111, 60-65.
Morris, N.M. & Udry, J.R. (1977) Study of the relationship between coitus and LH surge. Fert. Steril. 28, 440-442.
Moss, R.L. & Cooper, K.J. (1973) Temporal relationship of spontaneous and induced release of LH in normal cyclic rat. Endocrinology 92, 1748-1753.
Niswender, G.D. & Spies, H. (1973) Serum levels of luteinizing hormone, follicle stimulating hormone, and progesterone throughout the menstrual cycle of monkeys. J. clin Endocr. Metab. 31, 326-328.
Saxena, B.N., Poshyachinda, V. & Dusitin, N. (976) A study of the use of intermittent serum luteinizing hormone,progesterone, estradiol measurements for the detection of ovulation. Br.J. Obstet. Gynecol., 83, 660-664.
Schacter, S.& Singer, J.E. (1962) Cognitive social and physiological determinants of emotional state. Psychol. Rev. 69, 379-399
Stearns, E.L., Winter, J.S.D. & Faiman, C. (1973) Effects of coitus on gonadotropin, prolactin, and sex steroid levels in man. J. Clin. Endocr.Metab.37, 687-691
Treloar, A.E, Boynton, R.E., Behn, B. G. & Brown, B.W. (1967) Variation of the human menstrual cycle, through reproductive life I.J. Fert. 12, 77-127
Tsai, C.C. & Yen, S.S.C. (1971) The effect of ethinyl estradiol administration during early follicular phase of the cycle on the gonadotropin levels and ovarian function. J. Clin. Enocr. Metab. 33, 917-923
Tukey, J. (1978) Personal Communication. Princeton University.
Udry, J.R. & Morris, N.M. (1968) Distribution of coitus in the menstrual cycle. Nature 220, 593-596.
Vollman, R.F. (1977) The Menstrual Cycle. In Major Problems in Obstetrics and Gynecology, Vol. 7 Saunders, Philadelphia.
Weizenbaum, F. (1976) The effects of environmental lighting on sexual behavior and testosterone levels of female rats. Phys. Behav. 17, 909-913
Zemlan, F. & Adler, N.T. (1977) Hormonal control of female sexual behavior in the rat. Horm. Behav. 9, 345-357
Dr. Cutler's pheromone science has been "bottled" into vials of unscented fragrance additives that increase wearers' sexual attractiveness. AVAILABLE FOR PURCHASE HERE
Related Links:
Philadelphia Study on Athena Pheromone 10X for men
San Francisco State University study on Athena Pheromone 10:13
Boston Study on Postmenopausal women
About the Scientific Process of a double-blind study
Full Bibliography of Dr. Cutler's work
Hysterectomy: Before and After
Menopause: A Guide for Women and Those Who Love Them
More information on health over forty
More information on bone health as we age
More information on Dr. Cutler's pheromone discovery
Please visit our new guide to the medical school admissions process for premeds: Searching For Admission: The Smart PreMed Student's Guide to Applying for Medical School