Perimenopausal Sexuality
Winnifred B. Cutler, Ph.D., Celso-Ramon Garcia, M.D., Norma McCoy, Ph.D.
Archives of Sexual Behavior, Vol. 16, No. 3, 1987.
© Copyright 1987. Archives of Sexual Behavior.

This report covers (i) the results of an interview survey concerning sexual responses of 124 women during the perimenopause (menopause transition), and (ii) the results of 52 women’s prospective coital behavior records over 3 months with concomitant steroid analysis. Results indicated that (i) in general, the age group centering around 49 years appears not to be suffering from a particular sexual deficit in the dimensions of desire, response, or satisfaction in their sexual life, and (ii) a subset of women with especially low estradiol levels (<35 mg/pl) tends to have reduced coital activity.
KEY WORDS: menopause, sexuality; premenopausal transition; female sexuality; estrogens.
Intro:
Before the menopause occurs with its final cessation of menses (usually around the age of 51), a 7-year-period of premenopausal transition is common. This “perimenopausal” stage is characterized by changes in menstrual cycle length and bleeding pattern. Commonly, the cycles become unusually long (more than 33 days) or short (less than 26 days) (Treloar et al., 1967; Vollman, 1977). Additionally, changes often develop in the amount or duration of flow (Metcalf, 1979; Rutherford, 1978). Although some perimenopausal sequelae have been well characterized, including hot flashes, emotional lability, and the inception of osteoporosis, less well established are the patterns of sexual response and sexual behavior changes at this transition stage (Cutler, Garcia, and Edwards, 1983; Kupperman, Wetchler, and Blatt, 1959; McCoy and Davidson, 1985). Our survey evaluated the female perimenopause with respect to related steroid levels and prospectively gathered coital frequency.
Although there is a consensus within the biomedical literature that a declining capacity for sexual response coincides with the declining levels of estrogen occurring at menopause (see Cutler and Garcia, 1984), psychologists have published data showing no loss of arousal during this period (Neugarten et al. 1963; Luria and Meade, 1984). The particular age sample under review here – the transition stage – has not yet been reported systematically with respect to perceptions, prospective sexual behavior records, and concomitant steroid levels. Previous reports have shown that “weekly” coital frequency behavior is distinguished from less than weekly coital behavior: Women who have weekly sex with men show cycle length characteristics and basal body temperature (BBT) patterns reflective of a more fertile endocrine milieu. Weekly sex is also associated with an increased incidence of 29-day cycles and fertile type BBT charts in two populations: gynecologically mature young women (Cutler, Garcia, and Krieger, 1979a; Cutler et al., 1985), and infertile women (Cutler, Garcia, and Kreiger, 1979b). In perimenopausal women, weekly sex is associated with a reduced incidence of hot flashes (Cutler, McCoy, and Davidson, 1983; McCoy, Cutler, & Davidson, 1985).
Sample
One hundred fifty-five women volunteers responding to an announcement of a study of the menopausal transition formed the initial population of what was advertised as “The Stanford Menopause Study.” None of these women had had a hysterectomy or an ovariectomy; none was currently using hormonal therapy. The subjects responded to a brief television interview with the senior author or to similar local radio and newspaper announcements. Women from as far as 50 miles north of San Francisco to 30 miles south of Santa Cruz participated. The study was advertised as a “study of menopause” in an attempt to avoid preselectiong for “sexually open” women. The first interview was a nondirective open-ended questionnaire and interview process. For the second interview, 124 women (80%) returned to a personally convenient location away from Stanford; this interview provided the first set of data reported here. The third interview, approximately 3 months later, provided the second set of data and was conducted with 52 (34%) continuing subjects who were willing to come to Stanford for additional interviewing and to have plasma drawn for steroid assay as well as to keep prospective sexual behavior calendars. In a prior report, details of a subset of this population, who continued for a fourth interview, were presented (McCoy et al., 1985). The report focused on relationships of testosterone (T) and estradiol (E) to menopausal status as well as to hot flashes and sexual frequency. The present report focuses on the data collected by a questionnaire that evaluated a number of components in the larger sample.
Characteristics of the Sample:
In order to qualify for participation in the study, women had to have had a menstrual cycle within the last 2 years, but to have found an increasing irregularity either in length of cycle or pattern of flow. At the third interview, women reported varying intervals since the last menstrual period: Menses within the last 30 days, 56%; menses within the last 60 days, 20%; menses within the last 6 months, 9%; menses within the last 18 months, 5%; menses within the last 24 months, 5%.
The occurrence of hot flashes among their symptoms was reported by 71% of the women; 12% were experiencing night sweats as well. Only three women had used estrogen replacement therapy: two more than 2 months prior to entry into the study, one more than 18 months prior to entry. The educational level of the sample averaged 14.86 + 0.19 years (2 years college). The age distribution of the sample ranged between 33 and 56 years with the mean 48.67 + 0.34 years. The heaviest concentration of subjects ranged between 44 and 52 years (see Fig. 3). Menarche occurred at 12.7 + 0.12 years. Pregnancy rate averaged 3.24 + 0.16, live birth incidence 2.44 + 0.13, and length of marriage, on average, 20.8 years.
Method:
Interview 1 collected demographic data concerning age, medication, and surgical and reproductive system history, with particular attention directed toward defining the symptoms and sociomedical concerns of the women. The questionnaire employed open-ended questions with extensive space for anecdotal expressions of personal symptoms, questions, and concerns and provided the basis from which the objective questionnaire presented in this report was subsequently developed.
At the second interview, each subject completed a 31-item detailed questionnaire about sexual desire, response, and satisfaction, as well as a Kupperman questionnaire (Kupperman et al., 1959). The latter questionnaire allowed the derivation of Kupperman scores which are commonly used to reflect overall menopausal distress and are weighted to include hot flashes, night sweats, general nervousness, melancholy, and other well-documented menopausal symptoms.
The first and second interviews were conducted by 1 of 10 contemporaries of the subjects who served as volunteer research assistants under the direction of the senior author. The research assistants met with the senior author each week and conducted interviews in locations convenient for both the subjects and her research assistant. The third interview was conducted at Stanford where plasma was drawn. The sexual frequency data were collected prospectively using calendars which have been described in detail elsewhere (Cutler et al., 1979; Cutler et al., 1985). Blood samples were separated and the plasma frozen and later tested by radioimmunoassay for levels of estradiol and testosterone (see McCoy et al., 1985). Mean intraassay coefficients of variations were estradiol 9.7 and testosterone 7.8; the interassay coefficients were 13.5 and 6.9, respectively.
Questionnaire
What follows are (i) the results of an analysis of questionnaire data which dealt with sexual desire, sexual response, and sexual satisfaction in this perimenopausal age group, and (ii) a graphic array and analysis of coital frequency behavior pattern (weekly vs. less than weekly) with respect to age and estradiol levels.
A questionnaire was developed in 1979 and entitled “The 1980 Cutler/Davidson Menopause Project Questionnaire.” It was designed to investigate possible changes in a woman’s life prior to and during the menopause with specific reference to sexuality and menopausal distress symptoms. The first few questions evaluated the timing of a change in menstrual cycle habit and certainty about perception of when the timing of cycle patterns changed (i.e., change in onset, duration of flow, and quantity of menses). The next 25 questions followed a Likert scale procedure (with a 1 to 7 scale) that evaluated issues about sexuality including frequency of masturbation, frequency of intercourse, overall satisfaction with sexual life, orgasmic frequency during coital exposure, sexual pleasure, various measures of sexual response including lubrication and arousal, and measures of sex drive, including frequency of sexual thoughts and initiation of coitus. Other questions addressed potential distress including dyspareunia, satisfaction with partner, and partner’s satisfaction with subject. All questions had two parts: one relating to the last 30 days, the other to before the noticed change in menstrual cycling.
Results:
The questionnaire collected at the first of the interviews evaluated here (Interview 2) was initially arrayed for each question by K score. Then the distribution of data for one question versus another question was graphed in order to see the degree and potential relationship between variables. Twenty-three such combinations were arrayed. The first 16 compared a K score to various indices of sexual response. All yielded no apparent relationship. Menopausal distress scores in this sample were not reflective of sexual behavior. Seven comparisons were arrayed comparing two different aspects of sexuality: (i) orgasm frequency during intercourse vs. capacity to be aroused during sexual encounters; (ii) enjoyability at sexual encounters vs. arousability; (iii) dyspareunia incidence vs. sexual frequency; (iv) satisfaction with partner as a lover vs. satisfaction with partner as a friend; (v) dyspareunia vs. lubrication deficit at intercourse; (vi) dyspareunia vs. partner erectile problems; and (vii) frequency of sexual fantasies vs. capacity for arousal during sexual encounter.
In analyzing the questionnaire from interview 2, the desire for sexual activity was assessed by several different questions: frequency of sexual fantasy, recent change in fantasy, masturbation, and by the frequency of initiation of heterosexual behavior. As indicated by the data from the questionnaire, there was no recent change perceived by the subjects in the frequency of sexual fantasy nor in how this may or may not relate to menopausal distress (K score). The largest group (29%) reported their own occurrence of sexual fantasies to range between once a week and once a day. Approximately 20 % stated that they never had sexual fantasies. Less than 10 % had noted a significant change in sexual fantasy that coincided with their climacteric. The majority of women reported that they did not masturbate at all (74%). This frequency of self-reported masturbation is lower than that of reports in younger women (Cutler et al., 1979a). Sexual initiation appears to be predominantly the male partner’s behavior in this sample. There was no correlation between the frequency of masturbation and the frequency of sexual initiation.
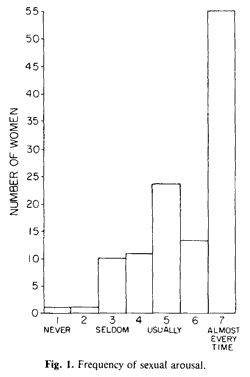
Questions of sexual responsiveness were addressed through questions dealing with arousal, vaginal lubrication response, dyspareunia, and association between orgasm and arousability. Figure 1 shows the response to the question: “How often during sex do you feel aroused or excited (for instance, increased heart beat/flushing/vaginal wetness/heavy breathing, etc.)?” With the majority this arousal occurs almost every time. A small portion of the women are of low arousability. The request, “estimate how often you had sexual thoughts or fantasies during the last month,” scored on a 7-point Likert scale from never to more than 10 times a day, was followed by a question, “Has this changed recently?” Twenty percent reported a decline in arousability that coincided with the recent history of the change in menstrual cycles; about 10% noted an increase.
The sexual lubrication response was evaluated as well. The question asked was “Do you currently suffer from lack of vaginal lubrication (wetness) during sex?” The overwhelming majority (86%) said they did not and for the 17 women who did report a vaginal lubrication deficit, their K score did not appear higher. Dyspareunia (painful intercourse) was rarely encountered; 82% reported the incidence as never or seldom.
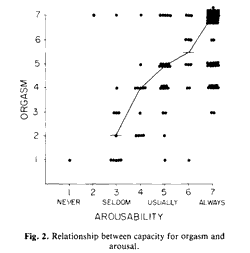
Figure 2 displays the data in response to the two questions: “How often during sex do you feel aroused or excited?” and “How often do you have an orgasm (climax) during sex?” There is a clear relationship between arousability and orgasm (r = 0.58, p < 0.001). Further, in this sample, the occurrence of orgasm as well as sexual arousal is common.
The calendar data were then analyzed in manners similar to previous reports (Cutler and Garcia, 1984; Cutler et al., 1986). As the calendar data and plasma sample were collected at the third interview, these data represent prospective data, the charting of which began at the completion of Interview 2, the questionnaire data.
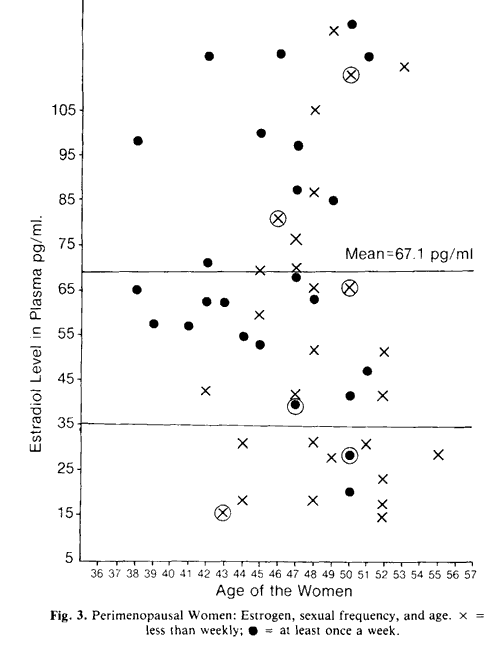
Figure 3 shows the estradiol level in each woman and indicates her age as well as her sexual frequency category. There is an apparent lack of relationship between age and estradiol level. For those women still menstruating, on the month of plasma drawing, samples were drawn during the first 5 days of the cycle – when estradiol is at its cyclic lowest. In Fig. 3, the line is drawn at 35 pg/ml because this is a commonly understood menopausal estradiol level and is lower than generally found on any of the days of the normal menstrual cycle (Cutler and Garcia, 1984). Although we have reported recently that “hormone levels were not significantly correlated with sexual activity over . . . the period of either interview,” a more significant hormone/behavior demonstration remained to be revealed, as shown here, because the data presentations (McCoy et al., 1985) assessed for a linear trend among the entire sample in coital behavior and hormone levels. The estradiol levels that were below 35 pg/ml show an increased proportion of less than weekly active women (z = 2.57, p < 0.005, Test of Proportions; Dixon and Massey 1969). Women whose estrogen levels reach this nadir are considered hormonally postmenopausal.
These results suggest that among the subset of women whose estradiol levels are clearly in the menopausal range, there is an increased proportion of women showing less than weekly sexual activity. This result is concordant with our report showing a “close association exists between . . . declining estradiol levels and declining frequency of intercourse during the perimenopause.” It also confirms a phenomenon we have reported recently in a sample of young (22-year-old) women. In that sample, women with less than weekly patterns of heterosexual behavior showed significantly lower estrogen levels, and women with estradiol levels below 40 pg/ml tended to be sexually inactive (Cutler et al., 1986).
Because of the nature of the study design, in which women were invited to participate if they had noticed a recent change in cycles or onset of symptoms, six of our subjects had stopped menstruating more than 12 months ago, as shown in “Characteristics of the Sample.” These six subjects did not represent any particular subsample of the data and are indicated by a circle around their data points on Fig. 3. They are rather well distributed with respect to estradiol level, age, and sex frequency.
Discussion
In spite of a commonly held general perspective of a decline in sexuality among menopausal women, this sample of perimenopausal women showed very little deficit. The capacity for sexual fantasy had not changed although the cycle pattern had.
There had been other evidence of estrogen declines in this group as revealed by hot flashes and changes in menstrual habit (Cutler et al., 1983; McCoy et al., 1985). The lubrication response likewise was not apparently affected nor was the potential for dyspareunia. This was not surprising since reports have shown that the loss of vaginal rugae, lubrication, and atrophy are noted as the later symptoms of menopause. This study was designed to evaluate women in the early perimenopausal stages. This sample cohort comprised predominantly white women with an approximately 2 years of college education. We have learned from the Gothenberg study, in which 800 women between the ages of 46 and 54 were studied, that a decline in sexual interest, capacity for orgasm, and coital frequency with each 5-year-period did occur. In that sample, the high social class of some of the women seemed to protect against this decline. In contrast, the lower social classes showed age-associated sexual deficits (Hallstrom, 1977). We must consider the possibility that other sociocultural groups demonstrate a different pattern that the group presented here.
Consideration should be directed to the often seen premise in medical books that purports that perimenopausal women have sexual deficits. The biomedical literature before 1983 suffered from a paucity of prospectively gathered data about the sexual habits of maturing women and, fortunately, this appears to be changing (Cutler, Garcia, and Edwards, 1983; Cutler and Garcia, 1984; McCoy et al., 1985; Leiblum et al., 1983).
In summary, the age group centering around 49 years appear not to be suffering from a particular sexual deficit in the dimensions of desire, response, or satisfaction with sexual life. However, about 25% of the women had very low (<35 pg/ml) estradiol concentrations and these women showed a substantially (and statistically significant) higher proportion of the “less than weekly” patterns of coital frequency.
ACKNOWLEDGEMENT
We thank Abba Krieger at the Wharton School of the University of Pennsylvania for performing the statistical analyses, and Dr. Julian Davidson at Stanford University, Department of Physiology, for performing the assays, reviewing the manuscript, and participating in developing the questionnaire.
Dr. Cutler's pheromone science has been "bottled" into vials of unscented fragrance additives that increase wearers' sexual attractiveness. AVAILABLE FOR PURCHASE HERE
REFERENCES
Cutler, W. B., and Garcia, C. R. (1984). Medical Management of Menopause and Premenopause:Its Endocrinologic Basis. J. B. Lippinicott, Philadelphia.
Cutler, W. B., Garcia, C. R., and Krieger, A. (1979a). Sexual behavior frequency and menstrual cycle length in mature premenopausal women. Psychoneuroendocrinology 4: 297-309.
Cutler, W. B., Garcia, C. R., and Kreiger, A. (1979b). Luteal phase defects: A possible relationship between short hypothermic phase and sporadic sexual behavior in women. Horm. Behav. 13: 214-218.
Cutler, W. B., Garcia, C. R., and Edwards, D. A. (1983). Menopause: A Guide for Women and the Men Who Love Them. W. W. Norton, New York.
Cutler, W. B., McCoy, N., and Davidson, J. M. (1983). Sexual behavior, steroids, and hot flashes are associated during the perimenopause. Neuroendocrinology 5: 185.
Cutler, W. B., Preti, G., Huggins, G. R., Erickson, B., and Garcia, C. R. (1985). Sexual behavior frequency and biphasic ovulatory type menstrual cycles. Physiol. Behav. 34: 5: 805-810.
Cutler, W. B., Garcia, C. R., Huggins, G. R., and Preti, G. (1986). Sexual behavior and steroid levels among gynecologically mature premenopausal women. Fertil. Steril. 45: 496-502.
Dixon, W. J., and Massey, S. J., Jr. (1969). Introduction to Statistical Analyses, 3rd ed. McGraw Hill, New York, pp. 249-250.
Hallstrom, T. (1977). Sexuality in the climacteric. Clin. Obstet. Gynecol. 4: 227-239.
Kupperman, H. S., Wetchler, B. B., and Blatt, M. H. G. (1959). Contemporary theory of the menopausal syndrome. J. Am. Med. Assoc. 171: 1627-1637.
Lieblum, S., Bachman, G., Kemman, E., Colburn, D., and Swartzman, L. (1983). Vaginal atrophy in the postmenopausal woman: The importance of sexual activity and hormones. J. Am. Med. Assoc. 249: 2195-2198.
Luria, X. and Meade, R. G. (1984). Sexuality and the middle-age women. In Baruch, G., and Brooks-Gunn, J., Women in Midlife. Plenum Press, New York.
McCoy, N. and Davidson, J. M. (1985). A longitudinal study of the effects of menopause on sexuality.Maturitas 7: 203-210.
McCoy, N., Cutler, W. B., and Davidson, J. M. (1985). Relationships among sexual behavior, hot flashes, & hormone levels in perimenopausal women. Arch. Sex. Behav. 14: 381-390.
Metcalf, M. G. (1979). Incidence of ovulatory cycles in women approaching the menopause.
J. Biosoc. Sci. 11: 39-48.
Neugarten, B. L., Wood, V., Kraines, R. J., and Loomis, B. (1963). Women’s attitudes towards the menopause. Vita Humana 6: 140-151.
Rutherford, A. M. (1978). The Menopause. New Zeal. Med. J. 87: 251-253.
Treloar, A. E., Boynton, R. E., Behn, D. G., and Brown, B. W. (1967). Variations of the human menstrual cycle through reproductive life. Int. J. Fertil. 12: 77-126.
Vollman, R. F. (1977). The menstrual cycle, Vol. 7. In Major Problems in Obstetrics and Gynecology. W. B. Saunders, Philadelphia.