Pheromones, Sexual Attractiveness and Quality of Life in Menopausal Women

Copyright ©2002. Parthenon Publishing; A CRC Press Company 2002
Journal: CLIMACTERIC The Journal Of the International Menopause Society June 2002
*Please click on bold print within text below for details and/or studies in full.
*Please click here for more on pheromones and books from Athena Institute
By Winnifred B. Cutler, Ph.D. and Elizabeth Genovese, M.D.
MESSAGE FROM DR. CUTLER: "What Dr. Elizabeth Genovese and I were invited by the Climacteric editors to do was to chronicle the important evolution of pheromone research; from pheromonal communication in animals to current discoveries including human sex attractant pheromones. Since Climacteric is the journal for the International Menopause Society, it was our intention to document the profound science of pheromones relevant to the sexuality and well-being of women in their menopausal years. In this article we address an unconventional scientific topic; sexual attractiveness.
We know that consensual intimate, affectionate behavior is good for your overall health. Investigations using topical pheromones demonstrated an increase in sexual attractiveness, and we describe those studies and relate them to a woman's romantic, and overall quality of, life at menopause.
The paper discusses rigorous scientific studies, however, non-medical readers will find it comprehensive and, we hope, beneficial." Winnifred B. Cutler
Dr. Cutler's pheromone science has been "bottled" into vials of unscented fragrance additives that increase wearers' sexual attractiveness. AVAILABLE FOR PURCHASE HERE
Below is the published paper in its entirety. For easy navigation, click on the tabs access each topic's section. Or click here to download a pdf of the paper.
- Abstract
- Biological Functions of Pheromones
- Source of Pheromones
- Synthesis of Pheromones
- Olfactory Mechanisms
- Intensity and Aging
- Potential use-Sex Dysfunction
Abstract:
Pheromones, and their effects, are reviewed with a special emphasis on their potential contribution to sexual attractiveness in the menopause. Key topics included are biological functions of pheromones in animals and humans, the source of pheromones in humans, the axillary extract studies that led to the independent synthesis of pheromones, olfactory mechanisms for mediating pheromones, and aging, attractiveness and sexual dysfunction.
Physical attractiveness is important for a better quality of life. Three separate, double-blind, placebo-controlled investigations, using the same protocol, all demonstrated that synthesized pheromone, topically applied, increased sexual attractiveness. If partners are available, sexual attractiveness can increase affectionate intimate behavior, which in turn, increases well-being and quality of life. More research is needed to address ways in which postmenopausal women can benefit from pheromones.
Introduction
In nature, sex pheromones are powerful agents driving reproduction and perpetuation of the species. Pheromones, and their effects, have been studied, although not extensively, in postmenopausal women
The term "pheromone" was first suggested in 1959 by biologists1 exploring a new observation: substances that had the chemical makeup of hormones but which were excreted elicited some behavior or developmental response in another individual of the same species. A considerable body of research has now confirmed that pheromones serve the reproductive life of species and are generally species-specific. Hence, the sex attractant pheromone of the wild boar is not an effective sex attractant for humans2.
Biological Functions of Pheromones
In humans, four behavioral classes of pheromones have been identified: opposite-sex attractants, male territorial markers or repellents that drive away sexual competitors, substances from babies also found in breast milk that facilitate the mother-infant bond, and male and female underarm essences involved in maintaining fertility and synchronization of menstrual cycles2,3.
We accept without question, that consensual affectionate behavior enhances well-being. While secreted hormones serve to maintain the self, excreted pheromones facilitate relationships with others2-4. Hormone replacement therapy enhances hormone-dependent, sexually responsive functions such as sexual motivation, arousal (lubrication and cognitive perception5, and orgasmic capacity. Synthesized pheromones may enhance the well-being of menopausal women by increasing their frequency of hugging, kissing and affectionate contact with men6.
The natural excretion of pheromones appears to be dependent on the fertile cycle of sex hormone production and to diminish with the decline of fertility , whether natural or surgically induced. The pioneering work with rhesus monkeys of psychiatrist Richard Michael, at Emory University, presents an unambiguous example. His group observed that ovariectomized rhesus monkeys were unable to attract sexual attention from males, despite actively soliciting through characteristic behavioral displays, in which they presented their attractive rear ends7.
In 1971, Michael and co-workers showed that vaginal smears from intact, sexually attractive, soliciting females, could be placed on the rear-ends of post-ovariectomized females to restore their capacity to attract male sexual attention8,9. He recognized that the substances had a 'pheromonal' action and named them "copulins." Subsequently, these monkey sex-attractants were tested in humans by smearing them nightly on the chests of married women, but without the anticipated positive results10. Species-specificity may explain this failure.
From 1975 to 1986, Cutler and her colleagues at the University of Pennsylvania performed a series of experiments in several hundred reproductive-aged women , investigating relationships between patterns of sexual behavior and fertility11. For at least 14 consecutive weeks, women recorded the occurrence of sexual behavior and menstrual onsets daily. Some studies also included daily basal body temperature records, and blood samples for sex hormone assay.
Subsequently, at Stanford University, similar data were gathered in 124 menstruating perimenopausal women. In composite, the studies showed that, compared with less sexually active women, those who engaged in at least one intimate heterosexual behavior per week also exhibited the following characteristics: more fertile basal body temperature pattern12, more normal menstrual cycle length (29.5 days)12,13, substantially higher levels of estrogen14-16, and, in the menopausal transition years, later onset of menopause and significantly lower incidence of hot flashes15,16.
Sporadic sexual behavior (feast and famine) was associated with more deleterious effects than abstinence, more aberrant cycles and the lowest levels of estrogen12,17,18. Sexual behavior during the week of menses was not necessary for these effects and was later found to be associated with increased risks for endometriosis19 and other excessive menstrual bleeding20.
As others replicated aspects of these findings21-23, it became clear that regular intimate exposure to a man, whether intercourse or genital stimulation in the presence of a man,was associated with positive health benefits. Solitary masturbation was not13. This distinction led Cutler and her colleagues to search for a human pheromone, investigating what a man provided a woman that she could not provide for herself.
Studies in animals suggested that sexual partner choice was mediated through pheromonal attraction, but, at that time, no such work had been published relating to humans. However, one intriguing study reported in 1980 that sweat from one woman's underarm, rubbed on another's upper lip three times a week for 4 months, caused the recipient's menstrual cycle to synchronized with the donor's cycle24. Subsequently, this donor's sweat was further tested on five other recipients in a placebo-controlled, but not blinded, trial with similar statistically significant results24.
Source of Pheromones in Humans: Axillary extract studies
In 1986 the first human, double-blind, placebo-controlled experiments provided strong support for the existence of an axillary source of pheromones in humans25,26. Reproductive-aged men and women, in ongoing heterosexual relationships, provided their underarm sweat, which was collected on sterile donor pads and, later, extracted, pooled and frozen. A year later, when these fluids were thawed and filtered to remove the odor and bacteria, an odorless clear fluid provided the first test substances , or 'essences', from humans.
Two double-blind, placebo-controlled, experiments with these substances demonstrated clear pheromonal effects. Women with aberrantly long or short cycle lengths received either the male essence or placebo. Pooled male essences, applied 3 times per week for 14 weeks to the skin under the nose on the women's upper lips, caused their menstrual cycle lengths to shift from aberrant to presumptively fertile (29.5 day) cycle lengths25. The aberrant cycles of women using placebo stayed aberrantly long or short. Hence, the male essence mimicked the behavioral effect of regular intimate exposure to a male, generating more presumptively fertile cycle lengths in women.
At the same time, women with presumptively fertile cycle lengths at baseline received either female essence or placebo. Female extracts from elite donors26 had been pooled by day of cycle to create a pooled day-1, a pooled day-3, a pooled day-5 extract and so on. These odorless substances were applied in sequence to the upper lip of naive women three times per week, for 14 weeks. The sequential female essence, but not the placebo, produced a shift in onset of the recipients' cycles that synchronized with the timing of the donor essences26. Thus, the applied elite female essence mimicked the behavioral effect that is now recognized; women in close contact tend to cycle together27,28.
In 1998, investigators at the University of Chicago further examined effects of female underarm essences on menstrual timing29. They concluded that daily application of pooled preovulatory essences shortened the follicular phase, advancing the onset of ovulation, while pooled postovulatory essences delayed ovulation. Although these effects were small, shifting ovulation time by an average of 1.7 days, they affected 68% of test subjects.
In 1995, Wedekind and colleagues32 provided additional support for the role of an axillary source of human pheromones in attraction. Young women were asked to score the pleasantness of used t-shirts of men in their mid 20's, most alike and most different from their own major histocompatability complex (MHC). The researchers reported that sweaty t-shirts were more attractive to women of different than of similar genetic make-up as reflected by their MHC. Two years later, these authors confirmed the findings in both men and women33.
These Wedekind studies provided further evidence that the underarm is an important site for sexual selection, and that genetic variations among individuals may serve to promote genetic variability in the immune systems of their offspring, perhaps to enhance the survival of the parent's genes.
Synthesis of Pheromones
Between 1986 and 1994, Cutler worked at the Athena Institute to synthesize two effective human sex-attractant pheromone formulas. With Dr. Norma McCoy at San Francisco State University and Dr. Erika Friedmann at Brooklyn College, she developed a rigorous, double-blind, placebo-controlled protocol to test the formulas. In 1994 in Philadelphia, 38 clean shaven, heterosexual men completed in the first double-blind experiment to test whether this putative male synthesized sex-attractant pheromone added to their preferred aftershave would "increase the romance in their lives".
Each man completed a daily checklist recording the occurrence of six behaviors that had occurred the day before, and faxed the calendar to the researchers each week. Results are given in TABLE 1, left column, Within 6 weeks of regular use, a significantly higher proportion of pheromone users than placebo users recorded increases over their average weekly baseline behaviors, in the average weekly frequencies of sexual intercourse and sleeping next to a romantic partner34.
Borderline effects were found for increases over baseline in petting/affection/kissing and informal dates. No difference occurred in average weekly frequency of masturbation, suggesting that these pheromones increased the sexual attractiveness of men to women rather than the sexual motivation of the wearer. Overall, 74% of the men who used the pheromone experienced an increase in at least one sociosexual behavior, an effect significantly higher than the 38% of placebo users who did so (p<0.01)34.
Professor Norma McCoy and Lisa Pitino tested the women's formula independently in 2000 in San Francisco35. They conducted a double blind, placebo controlled trial with 36 commuting college coeds with a mean age of 27.8 years ( range 19-47) who were not cohabiting or using an oral contraceptive. Data were collected over three consecutive menstrual cycles.
As in the men's study, the women recorded sexual behavior during a 2-week baseline. Afterwards they blindly selected placebo or the synthesized pheromone, added the vial to their own preferred perfume and then continued recording daily with weekly dispatch of their records. In order to provide six non-menstruating experimental weeks, the subjects provided daily data each week for three consecutive menstrual cycles.
Data were analyzed, as in the men's study, to provide a non-menstruating 2-week baseline and 6 week experimental period. Results revealed that a significantly higher proportion of pheromone than placebo users reported increases over their average weekly baseline behaviors in the frequencies of the four intimate sociosexual items recorded: sexual intercourse, sleeping next to a romantic partner, petting/affection/kissing, and formal, pre-scheduled dates.
TABLE 1: (click table to see larger)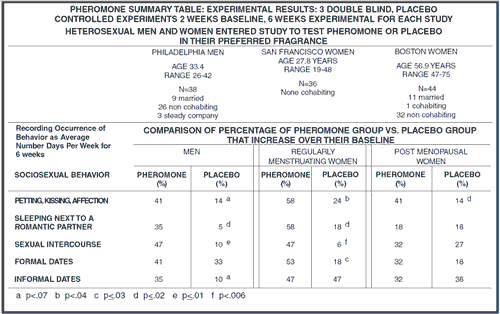
Table 1: Summary of experimental results from three double-blind, placebo-controlled trials: heterosexual men and women entered study to test pheromone or placebo in their preferred fragrance, comparing sexual behaviors during 2-week baseline and 6-week experimental periods, recording occurrence of behavior as average number of days per week.
No significant effect of pheromones was revealed for the two non-intimate sociosexual behaviors recorded: male approaches and informal dates, or for solitary sexual behavior, masturbation. Overall, 74% of the women who used the pheromone experienced an increase in at least three sociosexual behaviors, an effect significantly higher than the 24% of placebo users who did so (p<0.01).
While the San Francisco study was underway, Dr. Susan Rako, a Massachusetts psychiatrist and author specializing in postmenopausal sexuality issues, initiated an independent pheromone study. The Boston Postmenopausal Pheromonal Study investigated 44 intact (no hysterectomy or ovariectomy) postmenopausal women (mean age 56.9 years old) for a 2 week baseline and 6 week experimental period of daily recording of behavior.
Twenty-two women tested the synthesized pheromone and 22 others tested the placebo. Co-authoring with Dr. Joan Friebely, a researcher at Harvard University in the Psychiatry Department, Rako used the same behavioral questions and protocol as used in the San Francisco and Philadelphia Studies.
Focusing on the four intimate sociosexual behaviors that were increased in reproductive-aged women who used pheromone35, the Boston researchers found that 68% of the postmenopausal women who used pheromones experienced an increase in at least one behavior, an effect significantly higher that the 41% of placebo users who did so (p=0.03).
Within specific behaviors, a significantly higher proportion of pheromone than placebo users increased over their baseline behaviors in average weekly frequency of petting/affection and kissing6. However, the other sociosexual behaviors did not significantly increase.
Results in the menopausal group appear to be more modest than the results for men and women in their fertile years. Several explanations are possible. A reduced availability of male sexual partners occurs after 50. There is some evidence36 that with increasing postmenopausal age, there is a decreasing interest in sexual intercourse.
Moreover, in contrast with younger women, there is a reduced availability of male sexual partners for postmenopausal women. In fact, partner status at baseline (p=0.01) as well as pheromone use (p=0.03) were the two independent variables that significantly increased the likelihood that postmenopausal women would increase at least one intimate behavior during the 6-week experimental period. Postmenopausal women may require a longer experimental period, particularly if they need to find a partner, to bring about increases in more intimate sexual behaviors.
Olfactory Mechanisms for Mediating Pheromones.
Physiological mechanisms by which pheromones exert their demonstrated effects are poorly understood. Even in fungi, such as yeast, understanding of the mechanism of action is extremely limited , despite the identification of some interesting chemotactic mechanisms37.
In humans, pheromones appear to work through the olfactory system and without conscious awareness. They appear to be odorless. No discrete effects of scents on any behavioral change related to pheromones have been confirmed. For example, one study examined physiologic responses to erotic films with subjects simultaneously exposed to different fragrances38 ; another studied men and women who were blindfolded and presented scented strips of various substances, including hormones and control odors39. In both studies, only trivial and short-lived perceptual effects were obtained.
Young women at mid-cycle who sniffed androstenol recorded a transient suppression in aggressive mood, compared with those who sniffed placebo40. A reduction in deterioration of mood during tedious experiments while sniffing androgens and estrogens was also reported41,42.
Circulating androgens are positively correlated with mood and sexual desire in abnormally suppressed states during oral contraceptives use. Such hormone related mood and sexual motivation changes do not logically qualify as evidence of classical pheromonal effects. Anosmic individuals also have been evaluated44, to test the assumption that normal ability to perceive scent might be essential for adequate sexual life. No sexual deficits were identified. Hence, the absence of perception to known aromas, and the presence of responses to odorless substances, suggest that pheromones exert their effects independently of fragrance or other odors.
Gender differences in olfactory self-recognition exist; women have greater acuity45,46. For example, 59% of women but only 6% of men were able to recognize their own underarm odors from samples presented in blinded fashion46. It is doubtful that this gender difference in perception would affect the capacity to respond to sex attractants since the effective pheromone formulas used in the six studies6,25,26,30,34,35 discussed above were odorless.
Two anatomic olfactory sites in the nose are candidates for pheromonal receptivity. One is the olfactory epithelium, which carries neural impulses to the main olfactory bulb (MOB) enroute to cortical regions of the brain. The other is the vomeronasal organ (VNO), which carries impulses to the accessory olfactory bulb (AOB) enroute to subcortical regions of the brain.
The human VNO grows during gestation and is found as a bilateral structure in all normal adult nose47. Hormones mechanically pumped into the VNO inhibit gonadotropin releasing hormone and testosterone and provoke transient decreases in cardiac rate and respiratory frequency48. Sniffing such compounds activates the hypothalamic and ventromedial nuclie49,. Such drug-like mood-related effects are not classical pheromonal effects.
The MOB bypasses the VNO without any apparent communication. This is shown in mammalian maternal learning of an infant's odor and infant recognition of its mother in triggering the suckling reflex50. Stimulus access is via olfactory receptors, and the MOB has widespread projections to the neocortical processing areas of the brain in contrast to AOB which only projects subcortically. VNO destruction does not disrupt the maternal pheromonal behavior, suggesting that the neurophysiological connection is maintained solely via the main olfactory system50.
According to Brennan and Keverne's comprehensive review50, in a variety of animals the two systems have evolved separately to fulfill very different functions. The main olfactory system is able to make fine discriminations among an enormous variety of odor molecules, changing their associative values, and linking them to different behavioral outputs. In contrast, the accessory system (VNO) is highly specialized for the detection of a limited range of chemical messages, specific to that species, linked to relatively stereotyped behavioral and neuroendocrine outputs. Moreover, a recent investigation of the controversial claims49,51 for VNO involvement in mood changes when hormones were sniffed, investigators concluded that the VNO appeared neither necessary nor a sufficient pathway for these effects41.
In 2000, putative pheromone receptor gene expressed in human olfactory mucosa was reported for the first time52, establishing the first experimental basis for our deduction that sex-attractant pheromones are mediated in the olfactory epithelium.
Intensity
There is some evidence that more intense concentrations of pheromones become increasingly unattractive32,33,42. This maybe due to saturability of receptors or to some other as yet unexplored mechanism. At low doses, the effect of the combination of two bacterial pheromones on human neutrophils was additive for chemoattractiveness53. At higher doses the effects were not seen, suggesting that once receptors are saturated48,53 increasing the quantity does not increase the effectiveness.
Aging and Attractiveness: Will Pheromones Enhance Quality of Life in the Menopausal Years?
Physical attractiveness is important for a better quality of life. In young women, the perception that one is physically attractive appears to be important in positive sexual adjustment54. Perlini and colleagues55 demonstrated that younger and older judges of computer altered photos exhibited a clear attractiveness bias which can be accurately described as: "What is beautiful, is also more socially desirable and good".
Being judged attractive in the older years will lead to the perceptions by others that one is a better person than if one is not judged. Some evidence for a sex bias also exists in that older women are likely to perceive themselves as less attractive than they perceive older men56. Our Medline literature search yielded no references on sexual attractiveness in the menopause. A relative variation across cultures is documented in symptoms at menopause and their attribution to hormonal changes, fertility role changes, femininity, and status changes57, but the importance of "sexual attractiveness" remains unstudied, although widely appreciated. Sexual attractiveness is the ubiquitous lifeblood of pop culture.
In the USA, there is unanimity of public opinion, as expressed in media, that the more fertile-appearing the female, the more sexually attractive she is. Sex attractant pheromones may have the capacity to restore the subtle chemistry of this equation, much as hormonal replacement therapy helps to stem the tide of other menopausal declines.
Theoretically, five factors determine the sexual attractiveness of a woman:
1) Physical appearance and grooming;
2) Social skills - flirting, bodily grace, confidence;
3) Sexual motivation and interest in romantic contact and lovemaking;
4) Sexual skills,
5) Excretion of sex attractant pheromones.
Menopause declines occur in the physical, the motivational, and the pheromonal components. Because topically applied pheromones increase hugging, petting and affectionate contact with men, they have the potential to improve the quality of life for postmenopausal women who are experiencing an decline in attractiveness related to aging.
Sexual changes with age and hormonal diminution are well defined58. The McCoy female sexuality questionnaire (MFSQ) was developed to assess those aspects of feminine sexuality likely to be affected by declines in sex hormones59. The MFSQ has been used in seven recent studies and all show decreases in items ratings on sexuality as women progress through the menopausal transition and these ratings correlate with endocrine state. McCoy concluded that "no good evidence exists on the effect of menopause on sexual attractiveness; but the obvious hypothesis based on a literature review is that the effect is a negative one." In her view, a better understanding of attractiveness or "sexiness" in non-cycling women including the role of pheromones would undoubtedly shed further light on this issue58.
Perception counts. Studies of sexual arousal graphically measuring blood flow to the genitals reveal that blood flow contributes less to a woman's assessment of her own sexual excitement than does the woman's perception of her sexual arousal60. Hence perception appears to be more critical than objective scores. Similar findings in aging men by Davidson et al (cited in reference 11) affirm the importance of perception in the evaluation of patient needs. Women's satisfaction with their sexual relationships was also more closely associated with their marital adjustment and bore no relationship to age in a study at Oxford of 436 women61.
Sexual behavior changes as women progress from pre to postmenopause36. Findings show the frequency of sexual intercourse and orgasm declines in postmenopausal women
One of the most comprehensive studies of women's sexuality, by Dennerstein and her colleagues, evaluated 428 Australian born women aged 45-55 years at baseline with six annual assessments in the women's homes using selected questions form the MFSQ62. They reported that menopausal status had direct effects on 'feelings for their partner' , and the partner's sexual problems.
Recently63 investigators have moved toward developing a menopause specific "quality-of-life" questionnaire. A change in sexual desire, vaginal dryness during intercourse and avoidance of intimacy were each shown to be relevant to the "quality of life". Compared with the premenopausal years, 481 women in their postmenopausal years had a 10.6 fold higher risk for vasomotor problems, a 5.7 fold higher risk for physical impairments, a 3.5 fold higher risk for psychosocial impairments, and 3.2 fold higher risk for sexual impairments64. 'Being sexually active' was a protective factor against impairment in either physical or psychosocial conditions.
While the role of sexual attractiveness in promoting or maintaining sexual activity was not evaluated in any of the studies, such a role would seem likely. If partners are available, sexually attractive women can be expected to be more sexually active than are women who are not sexually attractive.
Potential for Pheromonal use in Sexual Dysfunction and After Hysterectomy
Controversy exists in the literature about the impact of hysterectomy on the sex life of women. Several studies have concluded that 1 and 2 years after hysterectomy, compared with 30 days before scheduled hysterectomy, sexual life has improved, for example in the frequency of intercourse65,66.
Recently we have challenged these conclusions arguing that the seeming improvements shown are biased by the choice of a baseline immediately preceding hysterectomy67. Choosing the sexual nadir in the lives of women awaiting hysterectomies, and showing improvement two years later does not support that it was hysterectomy that improved sex life. Rather, the reverse is supported by recent studies; sexual life is suppressed when hysterectomy is scheduled4,67.
Shifren and colleagues estimate that 29% of post hysterectomized women may suffer from sexual dysfunction; this conclusion was based on a survey inviting all patients whose surgery had occurred more than 1 year, but less than 6 years, earlier to participate in a 15-visit evaluation of sexual deficits68.
Research into female sexual dysfunction, like research on healthy female sexuality, has not yet incorporated an investigative vocabulary about attractiveness despite a seminal paper on the critical role of attractiveness in female sexuality published in 1976 69. For example, in 1999 Berman and co-workers70 reviewed the sexual dysfunction literature and the treatment options without ever mentioning sexual attractiveness. Heiman and Meston71 reviewed the Diagnostic and Statistical Manual of Mental Disorders (DSM IV) listing of sexual dysfunction standards, which also omitted any reference to sexual attractiveness. It is possible that the almost ubiquitous decline in sexual interest after the menopause is related to the decline in sexual attractiveness.
The evidence seems to suggest that pheromonal output varies among individuals and is highest during peak reproductive times such as at ovulation and during the fertile years11. Output diminishes with the loss of reproductive fertility. In the rhesus monkey, surgically induced loss of the reproductive hormones via ovariectomy and hysterectomy abolishes the natural secretion of sex-attractant pheromones.
Attractiveness can be restored with the topical application of species-specific pheromone or by replacing the missing reproductive hormones appropriately. In women, there is a suggestion that, after hysterectomy, a loss of sexual attractiveness may be commonly experienced.
Scholars at the University of Oklahoma, independently purchased and began testing the pheromone formula in postmenopausal women taking hormone replacement therapy who complained of a loss of attractiveness. They reported anecdotally72 - from their uncontrolled experiment - that about 70% of post-hysterectomized women using the topical pheromone described increased sexual activity within several weeks of regular use. Professor Clark Bundren terminated their study and recommended that patients simply use the pheromone product. There is a need for a proper double bind, placebo-controlled study.
The Future
Three separate double-blind, placebo-controlled investigations6,34,35 using the same protocol all demonstrated that a synthesized pheromone topically applied increased sexual attractiveness.
If partners are available, sexual attractiveness can increase affectionate intimate behavior, which in turn, increases well being and quality of life. In fact, regular sexual behavior is associated with physiological health.
Hence, in the future, physicians interested in fostering physical, psychological, and sexual health in their aging patients, may not only optimize function by prescribing sex hormones; they might also attempt to ensure that couples maintain their attractiveness to each other through the use of pheromones.
Summary of experimental results from three double-blind, placebo-controlled trials: heterosexual men and women entered study to test pheromone or placebo in their preferred fragrance, comparing sexual behaviors during 2-week baseline and 6-week experimental periods, recording occurrence of behavior as average number of days per week.
Percentage Recording Increase Over Baseline
Related Links:
Philadelphia Study on Athena Pheromone 10X for men
San Francisco State University study on Athena Pheromone 10:13
Boston Study on Postmenopausal women
About the Scientific Process of a double-blind study
Full Bibliography of Dr. Cutler's work
Menopause: A Guide for Women and Those Who Love Them
Hysterectomy: Before and After
More information on health over forty
More information on bone health as we age
More information on Dr. Cutler's pheromone discovery
Please visit our new guide to the medical school admissions process for premeds: Searching For Admission: The Smart PreMed Student's Guide to Applying for Medical School
" My research has consistently focused on what behavior a woman can engage in to increase her power, well-being, and vitality."
---Winnifred B. Cutler, Ph.D.
A portion of the profits from our book and pheromone sales helps to fund Athena's on-going research.